PhD In Business
Dr. Yohann Wittrant / CLINICNCELL in “PhD in Business”. A teaching module by IESF Auvergne and College des Ecoles Doctorales de Clermont Auvergne Université



Dr. Yohann Wittrant / CLINICNCELL in “PhD in Business”. A teaching module by IESF Auvergne and College des Ecoles Doctorales de Clermont Auvergne Université



Biomedicines 2024, 12, 2132
https://doi.org/10.3390/biomedicines12092132
Human Serum, Following Absorption of Fish Cartilage Hydrolysate, Promotes Dermal Fibroblast Healing through Anti-Inflammatory and Immunomodulatory Proteins
Julie Le Faouder, Aurélie Guého, Régis Lavigne, Fabien Wauquier, Line Boutin-Wittrant,
Elodie Bouvret, Emmanuelle Com, Yohann Wittrant* and Charles Pineau* (*co last-authors)



https://pubmed.ncbi.nlm.nih.gov/38543230/
Pharmaceutics. 2024 Feb 28;16(3):336. doi: 10.3390/pharmaceutics16030336.
Line Pourtau 1, Fabien Wauquier 2, Line Boutin-Wittrant 2, David Gaudout 1, Benjamin Moras 1, Adeline Vignault 1, Carole Vaysse 3, Tristan Richard 4 5, Arnaud Courtois 4 5, Stéphanie Krisa 4 5, Véronique Roux 6, Nicolas Macian 6, Gisèle Pickering 6, Yohann Wittrant 2 7 8



https://pubmed.ncbi.nlm.nih.gov/37735385/
Randomized Controlled Trial
BMC Musculoskelet Disord. 2023 Sep 21;24(1):748. doi:10.1186/s12891-023-06800-4.
Henrotin Yves 1 2, Julie Herman 3, Melanie Uebelhoer 3, Fabien Wauquier 4, Line Boutin-Wittrant 4, Anne-Françoise Donneau 5, Justine Monseur 5, Variance Mokam Fotso 3, Marie Duquenne 6, Mélanie Wagner 7, Elodie Bouvret 8, Bérénice Costes 3, Yohann Wittrant 9 10


Our expertise for human ex vivo clinical trials are now developped for animal care. Contact us for more details on these new clinical tools for veterinary approach. contact@clinicncell.com

https://pubmed.ncbi.nlm.nih.gov/37111121/
Nutrients 2023 Apr 14;15(8):1903. doi: 10.3390/nu15081903.
Circulating Human Metabolites Resulting from TOTUM-070 Absorption (a Plant-Based, Polyphenol-Rich Ingredient) Improve Lipid Metabolism in Human Hepatocytes: Lessons from an Original Ex Vivo Clinical Trial
Fabien Wauquier 1, Line Boutin-Wittrant 1, Stéphanie Krisa 2, Josep Valls 2 3, Cedric Langhi 4, Yolanda F Otero 4, Pascal Sirvent 4, Sébastien Peltier 5, Maxime Bargetto 5, Murielle Cazaubiel 5, Véronique Sapone 5, Annie Bouchard-Mercier 6, Véronique Roux 7, Nicolas Macian 7, Gisèle Pickering 7, Yohann Wittrant 8 9





clinicncell / clinic’n’cell
Les fondateurs de ClinicnCell sont ravis et honorés de rejoindre le Groupement des Industries de Santé et du Médicament de la Région Auvergne pour partager expériences, expertise et convivialité #GIMRA
https://www.gimra.info/les-actualites/
https://www.gimra.info/bienvenue-a-clinicncell/

https://www.ahajournals.org/doi/abs/10.1161/circ.146.suppl_1.11397?af=R

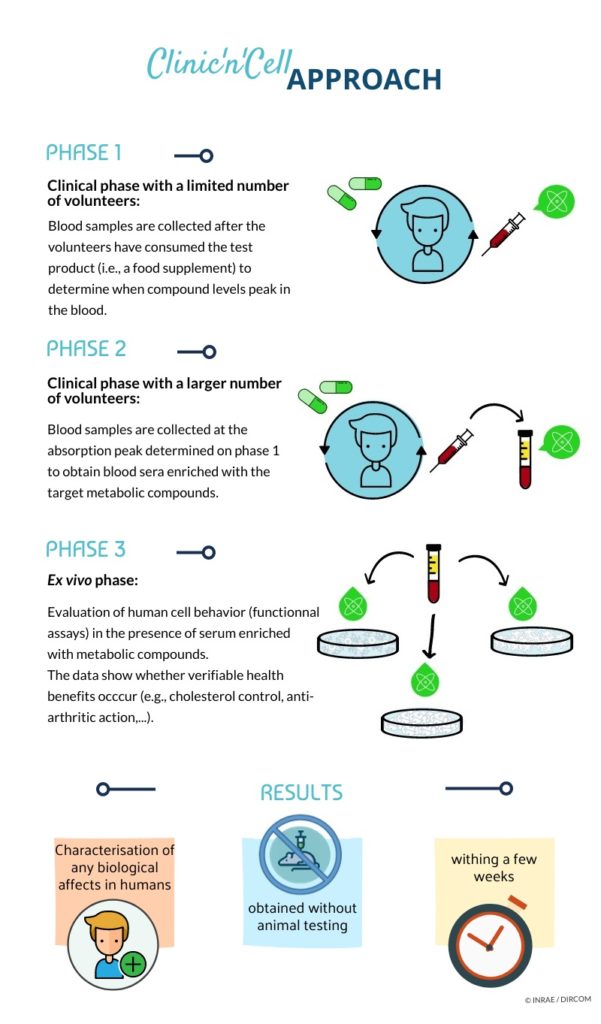
This clinical innovative approach provides robust human data regarding the health benefits of a product in faster, more affordable and more mechanistic ways (mode of action in humans) than any conventional clinical trial. Using human metabolites (circulating and bioavailable bioactives) this approach is more physiological than any in vitro test. The approach is also intended to be ethical since it allow to get rid of an animal experimentation. #clinicncell #clinic’n’cell
https://pubmed.ncbi.nlm.nih.gov/36501057/
Nutrients 2022 Nov 25;14(23):5027. doi: 10.3390/nu14235027
Benefits of Circulating Human Metabolites from Fish Cartilage Hydrolysate on Primary Human Dermal Fibroblasts, an Ex Vivo Clinical Investigation for Skin Health Applications.
Fabien Wauquier 1, Line Boutin-Wittrant 1, Elodie Bouvret 2, Julie Le Faouder 2, Véronique Roux 3, Nicolas Macian 3, Gisèle Pickering 3, Yohann Wittrant 4 5

ClinicnCell